Project summary
 The project aim is refinement of the web application intended for the maintenance of the regulatory reference documentation of the clinical diagnostic laboratory.
The project aim is refinement of the web application intended for the maintenance of the regulatory reference documentation of the clinical diagnostic laboratory.
Our team implemented new features for input, storage and operation of various kinds of data that are used in laboratory tests. BIRT technology was integrated to the application to provide the ability to generate and display various forms of reports.
The work of the team also included describing test cases and manual testing of the application after the tasks implementation.
Technologies
Java 6, XML, Maven, Spring Framework 3.2 (context, beans, web, security, ldap, orm, tx), Hibernate 4.2, c3p0, MySQL Connector, HSQL DB (embedded database for development), Jackson JSON mapper, JSP, JSTL, EL, SLF4J, LogBack, P6Spy, jUnit, JavaScript, ExtJS 4.2, JSON, BIRT
Screenshots
Project features
- Integration of BIRT technology into the system;
- Implementation of front-end part of the application using the ExtJS library;
- LDAP was used in parallel with the database in the system;
- The practice of Continuous Integration was applied in the project. Automatic builds and tests starts by commit, automatic deployment and usage of several branches of the application (production, development) were configured;
- Work in a mode of dynamic formation of requirements and changes of priorities by the customer;
- Interaction with the development team on the customer’s side;
- Setting up the role model for a large number of users;
- The development was carried out according to Scrum:
- daily status meetings;
- demo, summing up the results of the work.
Project results
- Functionality of the application was extended;
- BIRT technology has been integrated into the application: a number of reports and pages for inputting parameters for them have been prepared;
- The unit calculator with presence of alternative and basic units of measurement (which are converted in both directions) was developed;
- Improved work with the documents structure: the function of adding categories and subcategories of documents, the ability to move the document through the hierarchy were implemented;
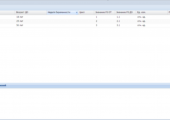
- The tabular form of displaying laboratory tests has been implemented;
- The search for laboratory tests has been improved; labeling of active laboratory studies was added;
- The user rights model has been improved.
Company’s achievements during the project
- Successfully and on time coped with all the tasks assigned;
- The project was carried out according to Scrum methodology, however it did not stop us from providing project plan to the customer for several stages ahead;
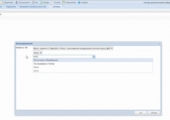
- The user form for selecting report parameters was implemented. The following options are available in the report parameters selection window:
- search for laboratory studies when choosing parameters by a combination of letters;
- possibility to add several laboratory studies;
- possibility to remove a mistakenly added laboratory study;
- Within the work on the application development, the differentiation of user access rights to the specified groups of documents are implemented. All nested documents groups that are descendants of the specified root groups inherited permission to access documents from their root group.
Articles created on the basis of project experience
Testimonials

"The main project task of JazzTeam engineers was to develop the new functionality of the web application intended for maintaining regulatory and reference documentation of a clinical diagnostic laboratory.
JazzTeam developers provided the final product development of remarkable quality and showed good teamwork. Quite often, we had to change the requirements and the priority for the tasks, but JazzTeam engineers showed flexibility and coherence while working in such situations."